
Bring Your Proven Formula,
We'll Scale It
Already have a winning supplement formula? Partner with a cGMP-certified manufacturer that specializes in high-volume scaling, cost optimization, and uncompromising quality control.
WHY TRANSFER?
Elevate Your Existing Product
Moving your production to our facility means more than just a new manufacturer—it's a strategic upgrade for your brand's supply chain.
Cost Optimization
Our global ingredient sourcing network often reduces raw material costs by 15-25% compared to smaller manufacturers.
Unmatched Quality ControlUnmatched Quality
Control
Every batch undergoes rigorous in-house and 3rd party testing to ensure your formula's integrity is never compromised.
Rapid Scaling
From 100,000 to 10,000,000+ units, our facility is engineered for high-volume production without lead-time delays.
Regulatory AssuranceRegulatory
Assurance
We review your existing formula for current FDA compliance and labeling requirements to mitigate brand risk.
Manufacturing Formats
We operate high-speed, automated production lines designed to handle the most complex formula specifications in these primary delivery formats.
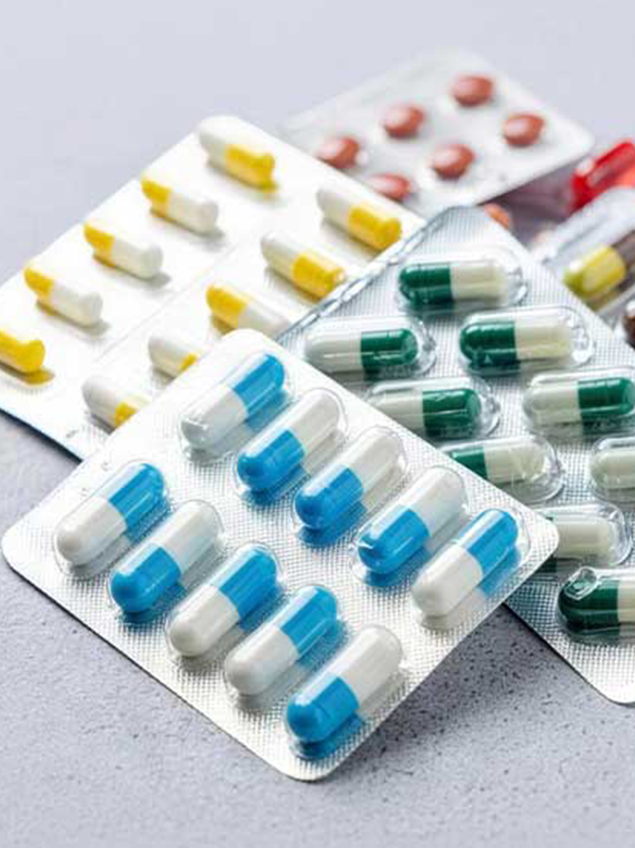
Capsules
Flexible formulation, easy to swallow
TECHNICAL SPECS
- Sizes: 00, 0, 1, 2, 3, 4
- Gelatin, Veggie & Delayed Release
- Clear, Colored, or Custom Print
- 10M+ Daily Production Capacity
BEST FOR
Botanicals, probiotics, oil-based nutrients, custom blends, sensitive ingredients
cGMP Certified
FDA REGISTERED FACILITY
Climate Controlled
24/7 MONITORING
Fully Automated
HIGH-PRECISION LINES
The NDA Promise
Your intellectual property is the lifeblood of your brand. We don't just sign NDAs; we build our entire infrastructure around the absolute security of your proprietary formulas.
Mutual NDA First
Legal protection is established before a single ingredient is shared.
100% IP Ownership
You retain full legal ownership of your formulas at all times.
Secure
Security Protocol
- Military-grade digital formula encryption
- Restricted 'Need-to-Know' R&D access
- Secure physical sample quarantine
- Biometric-controlled production areas
THE TRANSFER PATH
A Seamless Transition to
Superior Scale
Moving a proven formula is a critical decision. A precision-engineered transition framework designed to move your proven formulas to our high-capacity facility with zero disruption.
Formula Review
Submit your current spec sheet and COAs. Our R&D team reviews for manufacturing feasibility and optimization.
Ingredient Matching
We source identical or superior raw materials from our vetted global suppliers to match your exact profile.
Pilot Validation
We run a small-scale test batch to ensure the formula behaves perfectly on our high-speed equipment.
Full-Scale Launch
Once validated, we move to mass production with real-time quality monitoring and logistics support.
Formula Review
Submit your current spec sheet and COAs. Our R&D team reviews for manufacturing feasibility and optimization.
Ingredient Matching
We source identical or superior raw materials from our vetted global suppliers to match your exact profile.
Pilot Validation
We run a small-scale test batch to ensure the formula behaves perfectly on our high-speed equipment.
Full-Scale Launch
Once validated, we move to mass production with real-time quality monitoring and logistics support.
Transfer Checklist
To ensure a rapid and accurate transition, we recommend having the following documentation ready for our initial review.
Transfer Guide
Download our comprehensive technical migration whitepaper (PDF).
Ready to Improve Your Formula with a New Manufacturing Partner?
Submit your current capsule formulation for a confidential evaluation and receive a clear, competitive quotation backed by certified production standards.
Case Studies
R&D-Driven Formula Optimization with
Secure Manufacturing Protocols
Our formulation support goes beyond basic manufacturing. Through structured R&D validation and protected production systems, we ensure that proprietary supplement formulas are optimized, safeguarded, and ready for scalable commercial production.
Improving Bioavailability and Increasing Repeat Purchase Rate by 28%
The Challenge
The client's formula showed: · Low consumer-reported efficacy · Inconsistent dissolution performance · Poor differentiation in a competitive market
The Solution
RiverPharm's R&D team: · Replaced active ingredient with a more bioavailable form · Adjusted excipient ratio to improve dissolution · Conducted accelerated stability and dissolution testing · Performed pilot batch sensory evaluation
The Result
· Dissolution rate improved by 35% · Repeat purchase rate increased by 28% · SKU revenue grew by 32% in two quarters
Secure Formula Transfer with 100% IP Protection Compliance
The Challenge
During the client's previous collaboration with a manufacturer, insufficient confidentiality measures led to the leakage of its formulation. As a result, similar products entered the market, increasing competition from lower-priced alternatives with nearly identical positioning. This significantly weakened the client's product differentiation and profit margins, creating greater pressure on its market competitiveness.
The Solution
RiverPharm implemented: · Strict NDA enforcement across departments · Restricted-access formula coding system · Segmented production documentation · Controlled raw material sourcing · Internal audit trail for formula handling
The Result
· Zero IP breach incidents · Seamless production transfer within 90 days · Production volume scaled to 50,000 units per batch · Long-term manufacturing contract secured
Transfer Questions
A standard formula transfer typically takes 4-6 weeks from initial review to first production run. This includes ingredient sourcing validation and pilot batch testing.
Yes, absolutely. A mutual NDA is our standard operating procedure. We do not require you to share any proprietary blend information before legal protections are in place.
Yes. Our global supply chain network allows us to source nearly all branded and patented ingredients. In many cases, we can leverage our volume purchasing to get you better pricing on those exact ingredients.
Our typical MOQ for a formula transfer is 100,000 units (e.g., capsules) to ensure the efficiency of our automated lines. However, we evaluate each project individually based on formula complexity and long-term scaling potential.
Start Your Formula Review
Our R&D team will review your formula within 48 business hours.